Update for Apps Distributed in Texas
Update for Apps Distributed in Texas
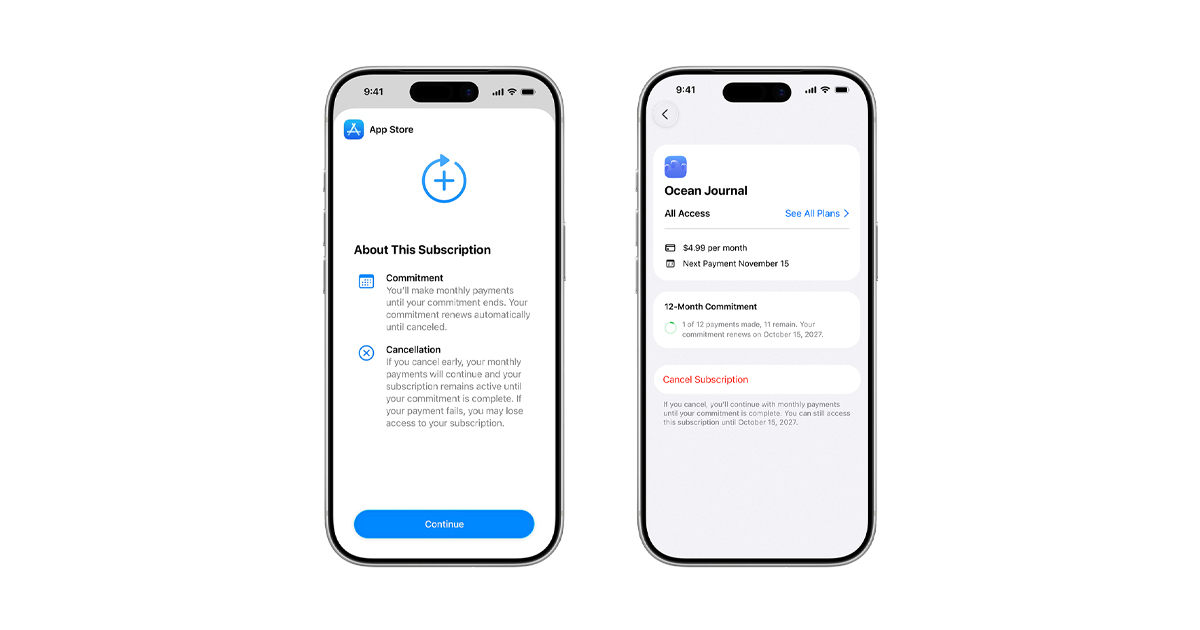
Due to a recent court ruling lifting an injunction on Texas law SB 2420, new Apple Accounts in Texas are now subject to the law, which introduced age assurance requirements for app marketplaces and developers.
Parents or guardians will also be able to revoke their consent for any app they previously approved for their child.
Developers can request age category data for these Apple Accounts through the Declared Age Range API.
For significant changes, developers should use the Significant Change API under the PermissionKit framework.
For the most up-to-date requirements and API references, see:Learn more about how you can provide age-appropriate experiences and safeguard privacy in your apps …
11 часов назад @ developer.apple.com infomate
infomate































![Apple kicks off new ‘Privacy on iPhone’ campaign promoting Safari [Video]](https://i0.wp.com/9to5mac.com/wp-content/uploads/sites/6/2026/06/safari-privacy-video.jpg?resize=1200%2C628&quality=82&strip=all&ssl=1)



![Apple permanently closing three US stores, here’s when [Updated]](https://i0.wp.com/9to5mac.com/wp-content/uploads/sites/6/2026/04/R285.png.jpeg?resize=1200%2C628&quality=82&strip=all&ssl=1)











































![Kodebits Day 34: String Interpolation [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/034.png)
![Kodebits: May 2026 in Review [FREE]](https://files.koenig.kodeco.com/uploads/2026/05/kodebits2_x_post_twitter.png)
![Swift SDK for Android [SUBSCRIBER]](https://assets.alexandria.kodeco.com/content_module/5e93a7da3f097c73d6d95a47ef37d21d63de50497f56e3b4b40cb94f3d464ce3/images/41ccc45d5e5f0264ce4e14dce5ddd7a5/original.png)
![Kodebits Day 31: Computed Property [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/031.png)
![Kodebits Day 28: Enum Associated Values [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/028.png)
![Kodebits Day 27: Optional Chaining [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/027.png)
![Kodebits Day 25: Capture List Value [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/025.png)
![Kodebits Day 24: Result Failure Path [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/024.png)
![Kodebits Day 23: Dictionary Defaults [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/023.png)
![Swift Metaprogramming: Writing Code that Inspects Itself [FREE]](https://files.koenig.kodeco.com/uploads/2026/05/swift_metaprogramming_x_post_twitter.png)
![Agentic Coding with Xcode [SUBSCRIBER]](https://assets.alexandria.kodeco.com/content_module/317d710efc13fed0f8c4fc2b9a35d0106a560a632af7bdf38ec5a2747b8b4c7e/images/15cceed62d8d6c72eb1389b600b80042/original.png)
![Kodebits Day 21: Result Map Value [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/021.png)
![Kodebits Day 18: Optional Chaining [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/018.png)
![Kodebits Day 17: Closure Reference Capture [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/017.png)
![Kodebits Day 15: Filter Then Sort [FREE]](https://files.koenig.kodeco.com/uploads/2026/02/015.png)